Compound Overview
Class of Compound:
Peptide
Mechanism of Action:
Liraglutide is known to activate the GLP-1R (glucagon-like peptide-1 receptors) in the body. It activates GLP-1R in the endocrine pancreas, which increases insulin secretion and reduces glucagon secretion. Liraglutide also activates GLP-1R in the central nervous system to promote satiety and in the gastrointestinal tract to slow gastric emptying.
Notable Studies:
- Efficacy and safety of liraglutide 3.0 mg in patients with overweight and obese with or without diabetes: a systematic review and meta-analysis
- Efficacy and safety of liraglutide for obesity and people who are overweight: a systematic review and meta-analysis of randomized controlled trials
- The effectiveness and safety of liraglutide in treating overweight/obese patients with polycystic ovary syndrome: a meta-analysis
Also Known As:
Victoza, Saxenda, Xultophy, NN2211, GTPL1133
Research Applications:
- Weight loss
- Metabolic syndrome
- PCOS
- Neuroprotection
Risks:
- Nausea and gastrointestinal upset
- Injection site reactions
- Tachycardia
- Potentially pancreatitis and neoplasias
- FDA approval limited to T2D and overweight/obese patients
Chemical Structure
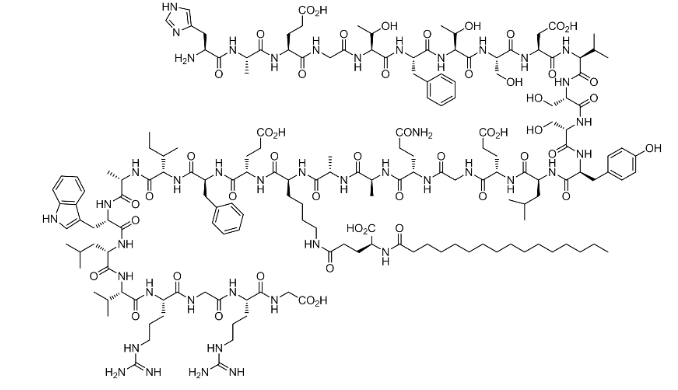
What is Liraglutide?
Liraglutide is a peptide that belongs to the class of drugs known as glucagon-like peptide-1 receptor agonists (GLP-1 RAs). It is a synthetic form of a naturally occurring hormone called glucagon-like peptide-1 (GLP-1) that helps blood sugar regulation [1].
Liraglutide is a long-acting GLP-1 RA that has a total of 31 amino acids and is 97% identical to the active form of the hormone GLP-1 (7-37).
Its structure is modified by the addition of palmitate to the amino acid lysine at position 26 of GLP-1 (7-37), which is added via a spacer glutamic acid residue [2].
The attached fatty acid moiety fosters binding to serum albumin, which delays renal excretion. This results in a half-life of 13 hours in humans.
Furthermore, lysine is replaced with arginine at position 34 of GLP-1 (7-37).
Liraglutide was discovered by the Danish pharmaceutical company Novo Nordisk in 1990 [3]. After extensive research, it was approved by the United States Food and Drug Administration (FDA) for the treatment of type 2 diabetes in 2010 [4].
In 2014, the peptide was also approved as a treatment in overweight or obese adults, while in 2020 it was also approved in adolescents for the same indications [5, 6].
Liraglutide is currently under investigation for its potential effects on obesity-related conditions such as polycystic ovary syndrome (PCOS), non-alcoholic fatty liver disease (NAFLD), and others.
Keep reading for more details on the exact mechanisms behind liraglutide benefits, side effects, dosage, and where to buy research-grade liraglutide online.

What Does Liraglutide Do?
Liraglutide works by mimicking the function of GLP-1 and activating its receptors in several organs, including the pancreas, the stomach, fat tissue, and the brain.
For reference, GLP-1 is produced in the intestines upon ingestion of carbohydrate-containing foods. It is an incretin that helps regulate blood sugar levels by stimulating insulin secretion in the pancreas. Scientists report that it can also be viewed as a neuropeptide as it is synthesized by neurons in the brainstem and subsequently secreted into the hypothalamus [7].
Similar to GLP-1, liraglutide stimulates insulin secretion by activating the beta cells in the pancreas and also activates the GLP-1R on the alpha cells, which inhibits glucagon secretion [8]. This makes it highly effective for glycemic control in type 2 diabetes.
Liraglutide can also activate the GLP-1R in the gastrointestinal system to decelerate gastric emptying, thereby curbing postprandial glucose surges. In fact, studies report a 23% reduction in gastric emptying speed within the first hour of administering liraglutide at 3.0mg compared to a placebo. Gastric emptying returns to its usual pace within 4 hours [9].
Scientists suggest that by interacting with the GLP-1R in adipose tissue, liraglutide may also reduce the release of triglycerides in the bloodstream and thereby increase insulin sensitivity [10].
Liraglutide activates the GLP-1R in the brain as well, which is thought to help reduce appetite and exert neuroprotective benefits [11]. The appetite-suppressing effects of liraglutide are also instrumental to its weight loss effect in overweight and obese individuals.
Liraglutide Benefits | Clinical Trials
Given that liraglutide has received FDA approval as an anti-diabetic and weight loss drug, extensive research has been conducted on its potential benefits and applications.
Here are the latest studies that shed light on the effects of liraglutide and how it may be employed by scientists for further research.
- Reduction of adipose tissue: One of the primary and approved uses of liraglutide is the treatment of overweight and obesity. Numerous clinical trials have shown that liraglutide can result in significant weight loss among such patients, regardless of whether they have diabetes. As of publication, the largest meta-analysis on long-term liraglutide therapy, which covered 6,000+ participants, revealed that the peptide causes 4-6% weight loss on average. This meta-analysis included only studies that lasted for more than 12 months and the liraglutide dosage was 3.0mg [12]. Further, one trial, conducted in obese patients with chronic obstructive pulmonary disease, reported an average weight loss of 8.2kg after 20 weeks of 3.0mg liraglutide daily administration [13].
- Improved glycemic control: Another primary and approved use of liraglutide is for glycemic control in type 2 diabetes. A systematic review of 43 randomized trials investigated the real-world clinical effectiveness of liraglutide for the treatment of type 2 diabetes mellitus. The review showed that liraglutide significantly reduces glycated hemoglobin (HbA1c) by 0.9%-2.2% within 6 months of treatment initiation. Moreover, liraglutide therapy was well tolerated with a low risk of hypoglycemia, and the beneficial effects were maintained for at least 12 months [14].
- Decreasing risk of type 2 diabetes complications: Some of the most common complications in type 2 diabetes are cardiovascular disease, kidney disease, and non-alcoholic fatty liver disease (NAFLD). Studies show that liraglutide reduces major adverse cardiovascular events in T2D patients with a history of myocardial infarction/stroke, as well as those with established atherosclerotic cardiovascular disease without myocardial infarction/stroke [15]. Further, a meta-analysis of 18 randomized controlled trials (RCTs) with 1,580 T2D patients reported that liraglutide also improves indicators of renal function such as urine albumin-creatinine ratio, serum creatinine, cystatin C, and several inflammatory markers [16].
- Increasing insulin sensitivity: Animal studies suggest that liraglutide improves insulin sensitivity and promotes fatty acid oxidation in insulin-sensitive tissues through the AMPK-Sirt-1 cell signaling pathway [17]. Liraglutide also induces brown adipocyte differentiation in skeletal muscle and displays anti-inflammatory effects, reducing the levels of circulating and hepatic cytokines. Clinical trials also suggest that liraglutide may improve insulin sensitivity, but more research is needed [18].
- Neuroprotective effects: Clinical research has aimed to evaluate the effect of liraglutide on cerebral glucose uptake, cognitive measures, cortical volume, and safety in individuals with mild to moderate Alzheimer's disease. In a phase 2 trial investigating the effects of liraglutide in mild Alzheimer's dementia, 204 participants received either liraglutide or placebo for 12 months, with the finding that the liraglutide-treated group had significantly better cognitive function and higher cortical volume compared to control [19]. This suggests that liraglutide may have neuroprotective properties, but more research is needed to confirm these findings.
Buy Liraglutide from our top-rated vendor...

Liraglutide Side Effects
Similar to other medications and research peptides, liraglutide has the potential to cause side effects. Studies in obese and diabetic patients have indicated that the most commonly reported side effects associated with liraglutide are symptoms of gastrointestinal upset such as [20]:
- Nausea
- Diarrhea
- Vomiting
- Constipation
According to Drugs.com, these side effects occur in 10-40% of participants during weight loss research. It's important to note that these reactions are transitory and can potentially be avoided by starting at a low dose of liraglutide, and gradually increasing the dose on a week-by-week basis before reaching the full dose. Other common side effects include increased heart rate and headache [21].
In addition, less than 10% of subjects may experience rashes, fatigue, other gastrointestinal problems, and reactions at the injection site, such as pain, bleeding, redness, swelling, or itching. Allergic reactions are rare.
Although uncommon, research has also revealed that liraglutide may cause more severe side effects, including pancreatitis, gallbladder disease, or cholestasis. Symptoms of such serious adverse reactions may include severe abdominal pain, vomiting, fever, and yellowing of the skin or eyes [22].
Moreover, ongoing research suggests that liraglutide may slightly increase the risk of certain cancers, such as pancreatic, breast, and thyroid cancer [23].
Is Liraglutide Safe?
As mentioned earlier, the FDA has granted injectable liraglutide approval for the management of several conditions, such as type 2 diabetes, overweight, and obesity in individuals aged 12 or older. The FDA issued these approvals only after there was sufficient data from multiple clinical trials for the safety and effectiveness of liraglutide.
Moreover, recent studies continue to explore the safety of liraglutide and report that the peptide is well-tolerated among most research subjects [24]. That being said, it is imperative that researchers consider the risks and benefits of administering liraglutide to test subjects for research purposes.
Furthermore, administering non-prescription liraglutide for any other purpose, such as general weight management, is not recommended. Non-prescription liraglutide should only be handled by qualified researchers or laboratory professionals, and it should never be self-administered for non-scientific purposes.
Liraglutide Dosage Calculator
The dosage of liraglutide in experimental settings depends on the research objective. For example, the maximum recommended dosage of liraglutide for fat loss is 3 mg/day, which can be reduced depending on individual tolerance [25].
It is important to note that at the beginning of each study, researchers should start with a low daily dose and increase the liraglutide dosage over the course of five weeks to prevent side effects. Here is a sample liraglutide weight loss protocol based on existing research [26]:
- Week 1: 0.6mg/day (starting dose)
- Week 2: 1.2mg/day
- Week 3: 1.8mg/day
- Week 4: 2.4mg/day
- Weeks 5+: 3.0mg/day (full dose)
For reference, the recommended liraglutide dosage for treating type 2 diabetes is 1.2-1.8mg/day [14].
Studies suggest that liraglutide may be administered for a long duration at the dose of 3.0mg/day, and the peptide should not be cycled. Instead, published research reveals that most common dosing protocols have a duration of 20-64 weeks [27].

Where to Buy Liraglutide Online? | 2024 Edition
When it comes to sourcing liraglutide for research purposes, we confidently recommend our preferred vendor.
PureRawz
There are several reasons why PureRawz is our top choice for purchasing research-grade liraglutide:
- Clean Research Chemicals: PureRawz has a comprehensive quality control program in place that includes vetting and supervising its production partners. They source their peptides from accredited facilities who adhere to Good Manufacturing Practices (GMP).
- Lab-Tested for Purity: PureRawz offers liraglutide that exceeds 99% purity. They rely on both in-house and third-party mass spectrometry testing to ensure purity and consistency in their products.
- Reliable Shipping: PureRawz offers secure online shopping and fast, reliable shipping. They provide affordable shipping fees and free shipping for orders above $100. They also ensure that their products are shipped promptly to prevent delays.
- Detailed Information: PureRawz provides detailed research summaries for each peptide on its website, including liraglutide. They also offer helpful tips on how to properly handle peptides.
With their commitment to quality assurance, product purity, and reliable shipping, PureRawz is the ideal choice for researchers looking to source liraglutide online.
As an added bonus, PureRawz is now offering a 15% discount. Just enter the following discount code at checkout:
PurePeptides
Buy Liraglutide from our top-rated vendor...

How to Reconstitute Liraglutide
Liraglutide for research purposes comes as a lyophilized powder, which must be reconstituted with bacteriostatic or sterile water.
To ensure safety during reconstitution, follow all standard precautions, such as applying alcohol wipes to clean the tops of both the peptide and sterile water vials, and using sterile needles and syringes to draw in the solvent and inject it into the powder.
When reconstituting the peptide solution, it is important to slowly inject the reconstituting liquid down the side of the vial. Allow the solution to dissolve naturally or use sonication.
Do not shake or tap the vial after reconstitution. Do not freeze, heat, or expose to direct sunlight.
Upon reconstitution, the stability of a peptide is contingent upon the type of solvent utilized. Microbial proliferation remains uninhibited in the presence of sterile water, rendering the peptide unsuitable for use within 24 hours, even under refrigeration.
In contrast, bacteriostatic water containing 0.9% benzyl alcohol exhibits no toxicity at these concentrations and acts as a microorganism growth suppressant.
It enables the peptide solution to remain viable for up to 4 weeks post-reconstitution when stored properly at a temperature range of 2 to 8 degrees C (36 to 46 degrees F).
Consult the specific product label for accurate instructions on dosage and storage requirements.
Liraglutide vs. Semaglutide
Semaglutide is also a peptide GLP-1 receptor agonist and FDA-approved medication used to treat type 2 diabetes, overweight, and obesity [28].
Both liraglutide and semaglutide work by stimulating the release of insulin and reducing the production of glucagon, which leads to lower blood sugar levels. Similar to liraglutide, semaglutide also interacts with the GLP-1 receptors in the brain to suppress appetite levels and help with weight loss.
Here are some differences between the two peptides:
- Dosage: As discussed, the maximum recommended dose for weight loss for liraglutide is 3.0mg, while for semaglutide, it is 2.4mg [25, 29].
- Administration Frequency: Liraglutide is administered once daily by subcutaneous injection, while semaglutide can be given once weekly by subcutaneous injection thanks to its considerably longer half-life of 6-8 days [30].
- Efficacy and side effects: In clinical trials, semaglutide has been shown to be more effective in reducing HbA1c levels than liraglutide. However, semaglutide has also been associated with a higher risk of nausea and vomiting compared to liraglutide [31].
Liraglutide vs. Dulaglutide
Dulaglutide (brand name Trulicity) is also a GLP-1 receptor agonist that is approved by the FDA for the treatment of type 2 diabetes. But unlike liraglutide, it has not been approved as a weight loss treatment for overweight and obese individuals [32].
Instead, dulaglutide has been approved for reducing major adverse cardiovascular events in adults with type 2 diabetes with established cardiovascular disease or multiple cardiovascular risk factors [33].
Here are some other differences between the two medications:
- Dosage: Liraglutide is available in doses of 0.6mg, 1.2mg, 1.8mg, and 3.0mg, while dulaglutide is available in doses of 0.75mg and 1.5mg.
- Administration Frequency: Both medications are administered by subcutaneous injection, but liraglutide is given once daily while dulaglutide is designed for once-weekly administration thanks to its half-life of approximately five days [34].
- Efficacy and side effects: In clinical trials, dulaglutide is slightly more effective than liraglutide in reducing HbA1c levels. Both peptides have similar safety profiles and risks. The most common adverse reactions with dulaglutide are gastrointestinal side effects such as nausea, vomiting, and diarrhea. However, dulaglutide has been associated with a lower incidence of nausea [35].
FAQ
Liraglutide | Reviews
Liraglutide is a GLP-1 receptor agonist that is approved for the treatment of type 2 diabetes and as a weight loss agent in overweight and obese individuals. Studies have shown that it is beneficial for reducing body weight and improving glycemic control in type 2 diabetes.
It is under active investigation for its potential in other areas of research, including for its potential neuroprotective effects.
Liraglutide can be legally purchased for research purposes by qualified professionals. The peptide is typically administered once daily via subcutaneous injection and exhibits a favorable safety profile to date.
For researchers interested in purchasing liraglutide, we recommend this trusted vendor of high-purity peptides.
References
- Müller, T. D., Finan, B., Bloom, S. R., D'Alessio, D., Drucker, D. J., Flatt, P. R., Fritsche, A., Gribble, F., Grill, H. J., Habener, J. F., Holst, J. J., Langhans, W., Meier, J. J., Nauck, M. A., Perez-Tilve, D., Pocai, A., Reimann, F., Sandoval, D. A., Schwartz, T. W., Seeley, R. J., … Tschöp, M. H. (2019). Glucagon-like peptide 1 (GLP-1). Molecular metabolism, 30, 72–130. https://doi.org/10.1016/j.molmet.2019.09.010
- Knudsen L. B. (2019). Inventing Liraglutide, a Glucagon-Like Peptide-1 Analogue, for the Treatment of Diabetes and Obesity. ACS pharmacology & translational science, 2(6), 468–484. https://doi.org/10.1021/acsptsci.9b00048
- Knudsen, L. B., & Lau, J. (2019). The Discovery and Development of Liraglutide and Semaglutide. Frontiers in endocrinology, 10, 155. https://doi.org/10.3389/fendo.2019.00155
- Iepsen, E. W., Torekov, S. S., & Holst, J. J. (2015). Liraglutide for Type 2 diabetes and obesity: a 2015 update. Expert review of cardiovascular therapy, 13(7), 753–767. https://doi.org/10.1586/14779072.2015.1054810
- Albaker, W., Al Sheikh, M., Albakr, A., Alkhafaji, D., Al Besher, E., & Al-Hariri, M. (2021). The Efficacy and Safety of Liraglutide 3.0 mg for Weight Management in Obese Non-Diabetic Saudi Outpatients. International journal of general medicine, 14, 8643–8650. https://doi.org/10.2147/IJGM.S336904
- Diene, G., Angulo, M., Hale, P. M., Jepsen, C. H., Hofman, P. L., Hokken-Koelega, A., Ramesh, C., Turan, S., & Tauber, M. (2022). Liraglutide for Weight Management in Children and Adolescents With Prader-Willi Syndrome and Obesity. The Journal of clinical endocrinology and metabolism, 108(1), 4–12. https://doi.org/10.1210/clinem/dgac549
- Cabou, C., & Burcelin, R. (2011). GLP-1, the gut-brain, and brain-periphery axes. The review of diabetic studies : RDS, 8(3), 418–431. https://doi.org/10.1900/RDS.2011.8.418
- Xu, X., Chen, J., Hu, L., Liang, M., Wang, X., Feng, S., Shen, J., & Luan, X. (2018). Liraglutide regulates the viability of pancreatic α-cells and pancreatic β-cells through cAMP-PKA signal pathway. Life sciences, 195, 87–94. https://doi.org/10.1016/j.lfs.2017.12.012
- van Can, J., Sloth, B., Jensen, C. B., Flint, A., Blaak, E. E., & Saris, W. H. (2014). Effects of the once-daily GLP-1 analog liraglutide on gastric emptying, glycemic parameters, appetite and energy metabolism in obese, non-diabetic adults. International journal of obesity (2005), 38(6), 784–793. https://doi.org/10.1038/ijo.2013.162
- Ejarque, M., Guerrero-Pérez, F., de la Morena, N., Casajoana, A., Virgili, N., López-Urdiales, R., Maymó-Masip, E., Pujol Gebelli, J., Garcia Ruiz de Gordejuela, A., Perez-Maraver, M., Pellitero, S., Fernández-Veledo, S., Vendrell, J., & Vilarrasa, N. (2019). Role of adipose tissue GLP-1R expression in metabolic improvement after bariatric surgery in patients with type 2 diabetes. Scientific reports, 9(1), 6274. https://doi.org/10.1038/s41598-019-42770-1
- Dailey, M. J., & Moran, T. H. (2013). Glucagon-like peptide 1 and appetite. Trends in endocrinology and metabolism: TEM, 24(2), 85–91. https://doi.org/10.1016/j.tem.2012.11.008
- Lin, Q., Xue, Y., Zou, H., Ruan, Z., Ung, C. O. L., & Hu, H. (2022). Efficacy and safety of liraglutide for obesity and people who are overweight: a systematic review and meta-analysis of randomized controlled trials. Expert review of clinical pharmacology, 15(12), 1461–1469. https://doi.org/10.1080/17512433.2022.2130760
- Altintas Dogan, A. D., Hilberg, O., Hess, S., Jensen, T. T., Bladbjerg, E. M., & Juhl, C. B. (2022). Respiratory Effects of Treatment with a Glucagon-Like Peptide-1 Receptor Agonist in Patients Suffering from Obesity and Chronic Obstructive Pulmonary Disease. International journal of chronic obstructive pulmonary disease, 17, 405–414. https://doi.org/10.2147/COPD.S350133
- Ostawal, A., Mocevic, E., Kragh, N., & Xu, W. (2016). Clinical Effectiveness of Liraglutide in Type 2 Diabetes Treatment in the Real-World Setting: A Systematic Literature Review. Diabetes therapy : research, treatment and education of diabetes and related disorders, 7(3), 411–438. https://doi.org/10.1007/s13300-016-0180-0
- Verma, S., Poulter, N. R., Bhatt, D. L., Bain, S. C., Buse, J. B., Leiter, L. A., Nauck, M. A., Pratley, R. E., Zinman, B., Ørsted, D. D., Monk Fries, T., Rasmussen, S., & Marso, S. P. (2018). Effects of Liraglutide on Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus With or Without History of Myocardial Infarction or Stroke. Circulation, 138(25), 2884–2894. https://doi.org/10.1161/CIRCULATIONAHA.118.034516
- Mali, N., Su, F., Ge, J., Fan, W. X., Zhang, J., & Ma, J. (2022). Efficacy of liraglutide in patients with diabetic nephropathy: a meta-analysis of randomized controlled trials. BMC endocrine disorders, 22(1), 93. https://doi.org/10.1186/s12902-022-01006-6
- Zhou, J. Y., Poudel, A., Welchko, R., Mekala, N., Chandramani-Shivalingappa, P., Rosca, M. G., & Li, L. (2019). Liraglutide improves insulin sensitivity in high fat diet induced diabetic mice through multiple pathways. European journal of pharmacology, 861, 172594. https://doi.org/10.1016/j.ejphar.2019.172594
- Anholm, C., Kumarathurai, P., Jürs, A., Pedersen, L. R., Nielsen, O. W., Kristiansen, O. P., Fenger, M., Holst, J. J., Madsbad, S., Sajadieh, A., & Haugaard, S. B. (2019). Liraglutide improves the beta-cell function without increasing insulin secretion during a mixed meal in patients, who exhibit well-controlled type 2 diabetes and coronary artery disease. Diabetology & metabolic syndrome, 11, 42. https://doi.org/10.1186/s13098-019-0438-6
- Edison, P., Femminella, G. D., Ritchie, C. W., Holmes, C., Walker, Z., Ridha, B. H., ... & Ballard, C. (2021). Evaluation of liraglutide in the treatment of Alzheimer's disease. Alzheimer's & Dementia, 17, e057848.
- Seo Y. G. (2021). Side Effects Associated with Liraglutide Treatment for Obesity as Well as Diabetes. Journal of obesity & metabolic syndrome, 30(1), 12–19. https://doi.org/10.7570/jomes20059
- Drugs.com (2023). Liraglutide Side Effects. https://www.drugs.com/sfx/liraglutide-side-effects.html
- Jensen, T. M., Saha, K., & Steinberg, W. M. (2015). Is there a link between liraglutide and pancreatitis? A post hoc review of pooled and patient-level data from completed liraglutide type 2 diabetes clinical trials. Diabetes care, 38(6), 1058–1066. https://doi.org/10.2337/dc13-1210
- Funch, D., Mortimer, K., Ziyadeh, N. J., D Seeger, J., Zhou, L., Ng, E., Ross, D., Major-Pedersen, A., Bosch-Traberg, H., Gydesen, H., & Dore, D. D. (2021). Risk of Thyroid Cancer Associated with Use of Liraglutide and Other Antidiabetic Drugs in a US Commercially Insured Population. Diabetes, metabolic syndrome and obesity : targets and therapy, 14, 2619–2629. https://doi.org/10.2147/DMSO.S305496
- Zhang, P., Liu, Y., Ren, Y., Bai, J., Zhang, G., & Cui, Y. (2019). The efficacy and safety of liraglutide in the obese, non-diabetic individuals: a systematic review and meta-analysis. African health sciences, 19(3), 2591–2599. https://doi.org/10.4314/ahs.v19i3.35
- Tilinca, M. C., Tiuca, R. A., Burlacu, A., & Varga, A. (2021). A 2021 Update on the Use of Liraglutide in the Modern Treatment of 'Diabesity': A Narrative Review. Medicina (Kaunas, Lithuania), 57(7), 669. https://doi.org/10.3390/medicina57070669
- Mehta, A., Marso, S. P., & Neeland, I. J. (2017). Liraglutide for weight management: a critical review of the evidence. Obesity science & practice, 3(1), 3–14. https://doi.org/10.1002/osp4.84
- Xie, Z., Yang, S., Deng, W., Li, J., & Chen, J. (2022). Efficacy and Safety of Liraglutide and Semaglutide on Weight Loss in People with Obesity or Overweight: A Systematic Review. Clinical epidemiology, 14, 1463–1476. https://doi.org/10.2147/CLEP.S391819
- Singh, G., Krauthamer, M., & Bjalme-Evans, M. (2022). Wegovy (semaglutide): a new weight loss drug for chronic weight management. Journal of investigative medicine : the official publication of the American Federation for Clinical Research, 70(1), 5–13. https://doi.org/10.1136/jim-2021-001952
- Villela, R., & Correa, R. (2022). Semaglutide 2.4 mg: the latest GLP-1RA approved for obesity. Journal of investigative medicine : the official publication of the American Federation for Clinical Research, 70(1), 3–4. https://doi.org/10.1136/jim-2021-002227
- Mahapatra, M. K., Karuppasamy, M., & Sahoo, B. M. (2022). Semaglutide, a glucagon like peptide-1 receptor agonist with cardiovascular benefits for management of type 2 diabetes. Reviews in endocrine & metabolic disorders, 23(3), 521–539. https://doi.org/10.1007/s11154-021-09699-1
- Deng, Y., Park, A., Zhu, L., Xie, W., & Pan, C. Q. (2022). Effect of semaglutide and liraglutide in individuals with obesity or overweight without diabetes: a systematic review. Therapeutic advances in chronic disease, 13, 20406223221108064. https://doi.org/10.1177/20406223221108064
- Fala L. (2015). Trulicity (Dulaglutide): A New GLP-1 Receptor Agonist Once-Weekly Subcutaneous Injection Approved for the Treatment of Patients with Type 2 Diabetes. American health & drug benefits, 8(Spec Feature), 131–134.
- Singh, R. B., Moshiri, M., Elmarghi, O., Elkilany, G., Fedacko, J., & Gushchina, Y. (2022). Glucagon Like Peptide Agonists as Therapy for Treatment of Cardiometabolic Diseases. World Heart Journal, 14(2), 145-156.
- Smith, L. L., Mosley, J. F., 2nd, Parke, C., Brown, J., Barris, L. S., & Phan, L. D. (2016). Dulaglutide (Trulicity): The Third Once-Weekly GLP-1 Agonist. P & T : a peer-reviewed journal for formulary management, 41(6), 357–360.
- Taheri, S., Saffaei, A., Amani, B., Akbarzadeh, A., Peiravian, F., & Yousefi, N. (2019). Efficacy and Safety of Dulaglutide Compared to Liraglutide: A Systematic Review and Meta-analysis in Patients with Type 2 Diabetes Mellitus. Iranian journal of pharmaceutical research : IJPR, 18(4), 2180–2190. https://doi.org/10.22037/ijpr.2019.14733.12619
