Compound Overview
Class of Compound:
Peptide
Mechanism of Action:
PT-141 is an agonist of the melanocortin receptors, with high affinity for MC4R, the stimulation of which increases sexual desire and causes sexual arousal in both men and women
Notable Studies:
Also Known As:
Bremelanotide, bremelanotide acetate, Rekynda, Vyleesi, CID9941379
Research Applications:
- Sexual funtion
- Male sexual arousal
- Anti-inflammatory action
Risks:
- Injection-related side effects
- Temporarily increases blood pressure
- Not FDA approved to treat men.
Chemical Structure
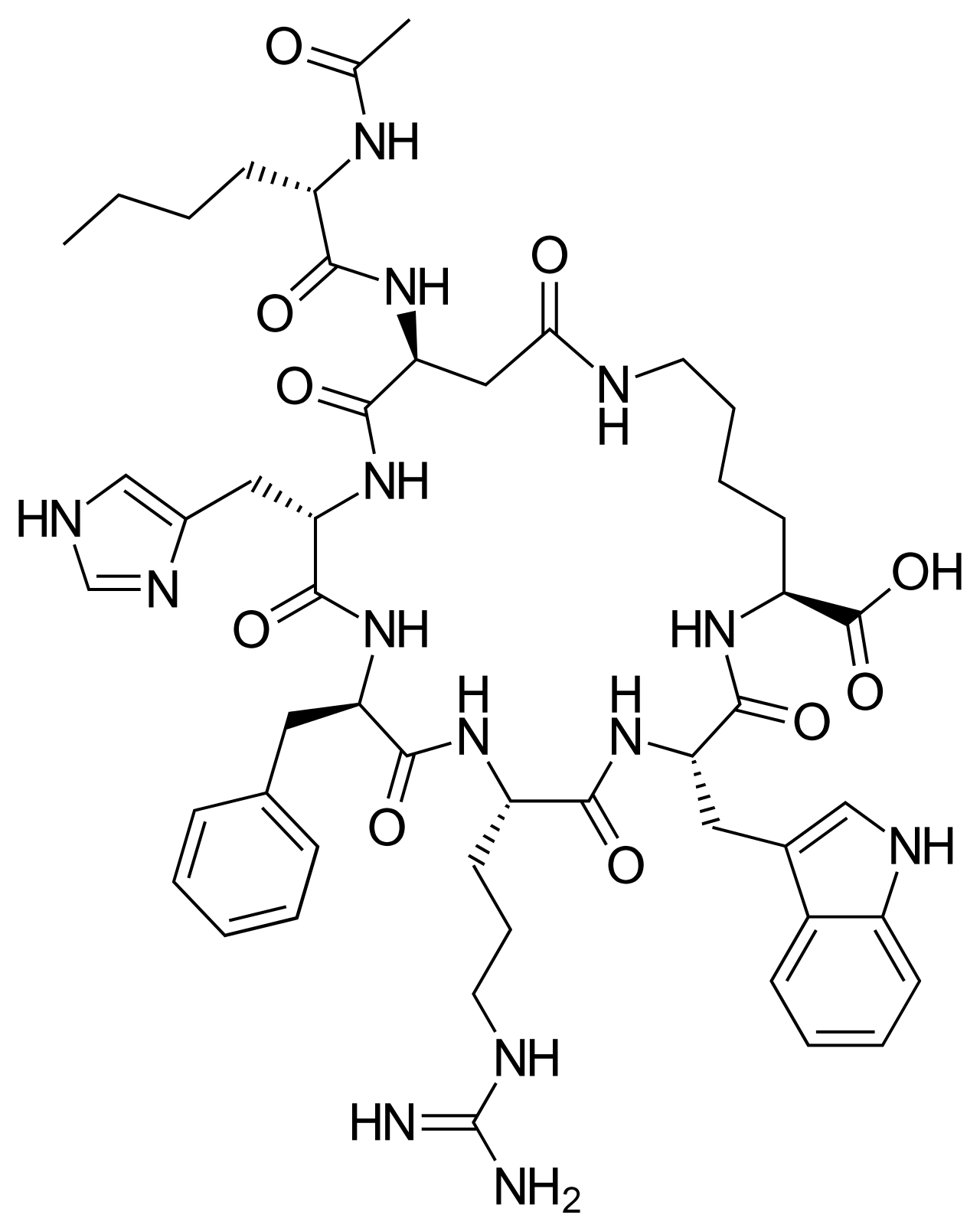
What is PT-141?
PT-141, also known as bremelanotide, is an FDA-approved treatment of hypoactive sexual desire disorder (HSDD) in women [1]. It is a modified synthetic derivative of alpha-melanocyte-stimulating hormone (α-MSH), a peptide of the melanocortin family that regulates skin pigmentation, feeding behavior, and sexual activity, among its other functions.
Back in the 1960s, researchers found that α-MSH caused caused sexual arousal when administered to rats, which eventually led to the development of α-MSH analogues like PT-141, melanotan I, and melanotan II.
In 2019, bremelanotide (PT-141) was approved in the United States as a therapy for premenopausal women with HSDD [2], and it is currently available under the brand name Vyleesi in auto-injection pens [3]. The manufacture and commercialization of Vyleesi in the United States are conducted by Palatin Technologies, the company that originally developed the drug [4, 5].
In addition to its role in treating HSDD in women, PT-141 has been extensively studied in a variety of contexts, including for its roles in treating erectile dysfunction (ED) and other forms of sexual dysfunction.

What Does PT-141 Do?
As an analogue of α-MSH, PT-141 acts on the melanocortin receptors (MCRs), which are found predominantly in the skin and central nervous system and cause an increase in pigmentation by activating melanocytes. However, MCRs are also associated with effects on sexual desire and appetite. PT-141 strongly engages MC1R, MC3R, and MC4R, the latter of which plays a role in modulating sexual desire [2].
Following a subcutaneous injection of PT-141, maximal levels occur after about one hour, with a range of 30 minutes to one hour [6]. In the realm of male sexual desire, this translates to a virtually on-demand erection.
PT-141 Benefits | Clinical Trials
PT-141 that is sold online is generally available only to qualified researchers and is not intended for human testing. However, since PT-141 is another name for the FDA-approved drug bremelanotide, the documented benefits and applications of bremelanotide are identical to those of PT-141:
Potential treatment of erectile dysfunction: While PT-141 is not indicated to treat erectile dysfunction (ED), several small-scale studies have found that it may be beneficial to male patients with ED who do not respond to sildenafil (Viagra).
A 2003 phase 1 randomized double-blind placebo-controlled trial involving 24 healthy male subjects without erectile dysfunction found that test subjects who received intranasal doses of PT-141 (4 to 20 mg) experienced “significantly increased duration of rigid erections of 140 minutes compared to 22 minutes in the placebo group” [6].
A 2004 study involving fewer than 400 patients found that doses of 1mg of PT-141 (bremelanotide) could produce a statistically significant “erectile response” [7].
Based on the above results, the safety and efficacy of PT-141 in test subjects with mild to moderate ED were investigated in a phase II study. Results showed a 3-fold increase in erectile activity following 20mg intranasal administration of PT-141 [8].
A 2008 study involving intranasal bremelanotide spray found that it could help men achieve an erection suitable for penetrative sex when administered 45 minutes prior to intercourse [9].
These findings suggest that PT-141 may act as an alternative ED treatment for subjects who do not respond to sildenafil, a potentiality that merits further research.
Treatment of HSDD in premenopausal women: As we have mentioned, bremelanotide is an FDA-approved treatment of low sexual desire in premenopausal women [4, 5].
While female sexual desire is not yet fully understood, evidence suggests that estradiol, testosterone, dopamine, norepinephrine, and serotonin all play a role.
PT-141 activates presynaptic MC4Rs on neurons and increases the amount of dopamine released [10]. Leading up to its approval, research on bremelanotide showed that premenopausal women with sexual dysfunction who were administered the peptide experienced more sexually satisfying events [11] and had less intercourse-related distress compared to placebo [12, 13].
General sexual dysfunction treatment: According to King et al., melanocortin agonists like PT-141 have “possible applications to the unmet medical needs of decreased sexual motivation and loss of libido” [14]. The authors cited the Molinoff (2003) study, where PT-141 was found to increase the rigidity of erections in test subjects with and without ED [6]. PT-141’s ability to trigger sexual desire in both men and women, independent of sexual stimulation, suggests that it holds potential as a treatment for a variety of sexual desire disorders.
Order PT-141 online from one of the top-rated vendors in the world...

PT-141 Side Effects
At the recommended dose of 1.75 mg, bremelanotide may produce the following common side effects [2]:
- Nausea (40% particularly with the first injection)
- Flushing (20%)
- Injection site reactions (13%)
- Headaches (11%)
Uncommon adverse reactions include [2]:
- Increases in blood pressure
- Severe nausea
- Vomiting
- Arthralgia
- Restless leg syndrome
- Local hyperpigmentation
Due to the risk of the peptide causing an increase in blood pressure and reduction in heart rate, it should not be administered to subjects who are [16]:
- Pregnant or suspected of being pregnant
- Suffering from cardiovascular disease
- Diagnosed with uncontrolled hypertension
In rare instances, PT-141 may lead to elevated serum enzyme levels, and in even rarer cases, cause acute liver damage [2].
Is PT-141 Safe?
When administered as a treatment of low sexual desire in women, bremelanotide has a favorable safety profile, having completed phase 3 clinical trials prior its being approved for use in the United States in 2019 [15].
In the field of male sexual desire, preliminary trials have established both the safety and efficacy of PT-141, showing that it is a potent initiator of erection with minimal side effects [14]. It must be reiterated that PT-141 is not approved to treat male sexual dysfunction at this time, and that PT-141 sold online is generally available for educational or scientific research only.
PT-141 Dosage Calculator
As a treatment of low sexual desire in women, bremelanotide is typically administered subcutaneously via self-injection. The most common dosage is 1.75 mg, roughly 45 minutes prior to anticipated sexual activity [2]. Patients with HSDD who are prescribed bremelanotide are advised to self-administer it no more than once per day, for a maximum of 8 times per month [13]. Those who experience adverse reactions to bremelanotide can reduce the dosage. Research has shown that doses of just 1.25 mg can still produce an effective response [11].
Studies involving male subjects with or without ED have tested subcutaneous injections of PT-141 ranging from 1 to 10 mg, showing that PT-141 can produce statistically significant results at doses exceeding 1 mg [7] and dose-dependent improvements in erectile function at doses up to 20 mg [14]. Research suggests a starting dose of 1 mg as a treatment of erectile dysfunction.
Where To Buy PT-141 Online? | 2024 Edition
Researchers interested in sexual health may be curious about where to buy PT-141 online.
Having tested multiple online peptide vendors and ranked them based on factors like peptide quality, shipping times, and customer support, here is our preferred PT-141 source:
Limitless Life
Limitless Life is the premier source of research-grade PT-141 at reasonable prices, and we recommend them for their:
- Third-Party Tested PT-141: All Limitless Life compounds undergo HPLC-MS analysis by a third-party laboratory to ensure identity, purity, and potency, making this vendor’s products some of the best on the market.
- Secure Website: Limitless Life offers a secure, fast website that protects customer data. We did not have any worries about our data being exposed or compromised when we ordered from this vendor.
- Impressive Support and Service: Our team was impressed by the support and service offered by Limitless Life. They were highly responsive by phone and email, which kept us in the loop at every stage of the buying process.
- Rapid Shipping Options: Limitless Life offers some of the best shipping options in the research peptides industry. They have two-day FedEx shipping standard on domestic orders, and also offer affordable shipping insurance.
- International Shipping: Limitless Life also offers international shipping that is timely and cost effective.
Not only that, but Limitless Life also offers a unique and powerful PT-141 nasal spray that is research-grade.
Furthermore, this research peptides vendor offers great pricing. This is compounded by them offering Peptides.org an impressive 10% off the next order for all qualified researchers.
Just click the button below and add this code:
peptidesorg10
Buy PT-141 from our top-rated vendor...

Bacteriostatic Water and PT-141
Experts advise that proper peptide handling requires a standard set of lab supplies.
To correctly reconstitute, administer, and store PT-141, your lab should be equipped with sterile vials, insulin needs, and bacteriostatic water, among other required tools.
Don’t let the hassle of gathering all the necessary supplies put a damper on the research!
While it can be time-intensive, these are some of the lab suppliers and materials every researcher will require:
- Bacteriostatic Water
- Insulin Syringes
- Large Needles + Syringes
- Alcohol Prep Pads
- Sterile Empty Glass Vials
FAQ
PT-141 | Reviews
PT-141 is a research peptide that has been clinically proven to increase sexual desire in both animal and human studies.
With a favorable safety profile, bremelanotide (PT-141) is indicated to treat low sexual desire in women in the United States. PT-141 has also been extensively tested, with promising results, as a potential treatment of erectile dysfunction.
Researchers looking to further study PT-141 are strongly encouraged to consider our top-rated supplier — a reliable, trustworthy vendor that ships research-grade PT-141 worldwide.

References
- National Center for Biotechnology Information (2021). PubChem Substance Record for SID 319341034, 189691-06-3, Source: ToxPlanet. Retrieved October 21, 2021 from https://pubchem.ncbi.nlm.nih.gov/substance/319341034.
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012–. Bremelanotide. 2021 Aug 20. PMID: 34436837.
- Dhillon S, Keam SJ. Bremelanotide: First Approval. Drugs. 2019 Sep;79(14):1599-1606. doi: 10.1007/s40265-019-01187-w. PMID: 31429064.
- Mayer D, Lynch SE. Bremelanotide: New Drug Approved for Treating Hypoactive Sexual Desire Disorder. Ann Pharmacother. 2020 Jul;54(7):684-690. doi: 10.1177/1060028019899152. Epub 2020 Jan 1. PMID: 31893927.
- Bremelanotide (Vyleesi) for hypoactive sexual desire disorder. Med Lett Drugs Ther. 2019 Jul 29;61(1577):114-116. PMID: 31381550.
- Molinoff PB, Shadiack AM, Earle D, Diamond LE, Quon CY. PT-141: a melanocortin agonist for the treatment of sexual dysfunction. Ann N Y Acad Sci. 2003 Jun;994:96-102. doi: 10.1111/j.1749-6632.2003.tb03167.x. PMID: 12851303.
- Rosen RC, Diamond LE, Earle DC, Shadiack AM, Molinoff PB. Evaluation of the safety, pharmacokinetics and pharmacodynamic effects of subcutaneously administered PT-141, a melanocortin receptor agonist, in healthy male subjects and in patients with an inadequate response to Viagra. Int J Impot Res. 2004 Apr;16(2):135-42. doi: 10.1038/sj.ijir.3901200. PMID: 14999221.
- Diamond LE, Earle DC, Rosen RC, Willett MS, Molinoff PB. Double-blind, placebo-controlled evaluation of the safety, pharmacokinetic properties and pharmacodynamic effects of intranasal PT-141, a melanocortin receptor agonist, in healthy males and patients with mild-to-moderate erectile dysfunction. Int J Impot Res. 2004 Feb;16(1):51-9. doi: 10.1038/sj.ijir.3901139. PMID: 14963471.
- Safarinejad MR, Hosseini SY. Salvage of sildenafil failures with bremelanotide: a randomized, double-blind, placebo controlled study. J Urol. 2008 Mar;179(3):1066-71. doi: 10.1016/j.juro.2007.10.063. Epub 2008 Jan 18. PMID: 18206919.
- Pfaus JG, Sadiq A, Spana C, Clayton AH. The neurobiology of bremelanotide for the treatment of hypoactive sexual desire disorder in premenopausal women. CNS Spectr. 2021 Jan 18:1-9. doi: 10.1017/S109285292100002X. Epub ahead of print. PMID: 33455598.
- Clayton, A. H., Althof, S. E., Kingsberg, S., DeRogatis, L. R., Kroll, R., Goldstein, I., Kaminetsky, J., Spana, C., Lucas, J., Jordan, R., & Portman, D. J. (2016). Bremelanotide for female sexual dysfunctions in premenopausal women: a randomized, placebo-controlled dose-finding trial. Women's health (London, England), 12(3), 325–337. https://doi.org/10.2217/whe-2016-0018
- Kingsberg SA, Clayton AH, Portman D, Williams LA, Krop J, Jordan R, Lucas J, Simon JA. Bremelanotide for the Treatment of Hypoactive Sexual Desire Disorder: Two Randomized Phase 3 Trials. Obstet Gynecol. 2019 Nov;134(5):899-908. doi: 10.1097/AOG.0000000000003500. PMID: 31599840; PMCID: PMC6819021.
- (2021). Retrieved 10 October 2021, from https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/210557s000lbl.pdf
- King SH, Mayorov AV, Balse-Srinivasan P, Hruby VJ, Vanderah TW, Wessells H. Melanocortin receptors, melanotropic peptides and penile erection. Curr Top Med Chem. 2007;7(11):1098-1106. PMID: 17584130; PMCID: PMC2694735.
- Simon JA, Kingsberg SA, Portman D, Williams LA, Krop J, Jordan R, Lucas J, Clayton AH. Long-Term Safety and Efficacy of Bremelanotide for Hypoactive Sexual Desire Disorder. Obstet Gynecol. 2019 Nov;134(5):909-917. doi: 10.1097/AOG.0000000000003514. PMID: 31599847; PMCID: PMC6819023.
- Althof S, Derogatis LR, Greenberg S, Clayton AH, Jordan R, Lucas J, Spana C. Responder Analyses from a Phase 2b Dose-Ranging Study of Bremelanotide. J Sex Med. 2019 Aug;16(8):1226-1235. doi: 10.1016/j.jsxm.2019.05.012. Epub 2019 Jul 2. PMID: 31277966.
